Implantable Biomaterials Choices That Raise Long-Term Risk
Not all implantable biomaterials behave the same after implantation.
A poor material choice can trigger inflammation, loosening, corrosion, delayed healing, or revision surgery years later.
That is why understanding implantable biomaterials matters before choosing an orthopedic, cardiovascular, spinal, dental, or soft tissue implant.
This guide explains which implantable biomaterials may raise long-term risk, how to compare them, and what questions help support safer decisions.
What are implantable biomaterials, and why do they affect long-term outcomes?
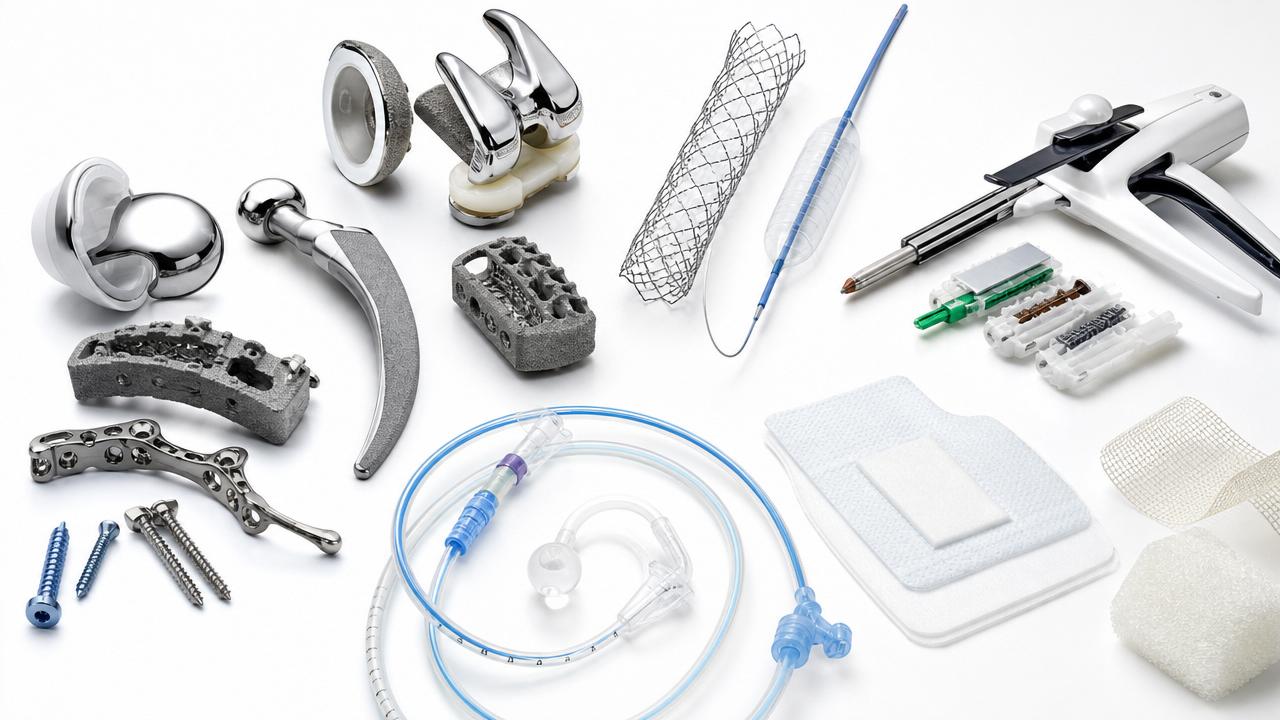
Implantable biomaterials are engineered substances placed inside the body to restore structure, function, or healing support.
Common examples include titanium alloys, cobalt-chromium, stainless steel, PEEK, ceramics, hydrogels, and biodegradable polymers.
Their job is not only mechanical.
They must also remain stable in blood, bone, soft tissue, and immune environments over long periods.
When implantable biomaterials mismatch the clinical need, problems can appear slowly rather than immediately.
Examples include wear debris, ion release, fibrotic encapsulation, weak osseointegration, thrombosis, or biofilm-related infection.
Long-term safety depends on several linked factors:
- Biocompatibility with local tissue
- Mechanical strength under real body loads
- Corrosion and wear resistance
- Surface design and tissue integration
- Sterility, cleanliness, and manufacturing precision
In short, implantable biomaterials influence both the first weeks of healing and the next ten or twenty years of performance.
Which implantable biomaterials choices most often raise long-term risk?
Risk does not come from one material alone.
It usually comes from choosing the wrong material for a specific anatomy, load pattern, or tissue environment.
1. Metals with corrosion or ion release concerns
Some metal implants perform well for decades, but certain combinations may release metal ions under friction or corrosion stress.
Cobalt-chromium systems can be durable, yet wear particles may increase inflammatory reactions in selected cases.
Nickel-containing materials may also be problematic for patients with metal sensitivity.
2. Polymers that wear, crack, or age poorly
Medical polymers are valuable because they are light, flexible, and imaging-friendly.
However, lower-grade polymer design may lead to fatigue cracks, deformation, or particle generation over time.
This matters in spinal cages, catheters, and some tissue-contact devices.
3. Ceramics that are stable but brittle
Ceramics can offer excellent wear resistance and good biological response.
Yet brittleness remains a concern where impact, edge loading, or unexpected stress concentration is possible.
4. Biodegradable materials with uncertain degradation behavior
Bioabsorbable implantable biomaterials can reduce permanent foreign-body burden.
Still, if degradation is too fast, too slow, or uneven, local acidity and weak structural support may follow.
5. Smooth or poorly engineered surfaces
Sometimes the core material is acceptable, but the surface fails to support integration.
A poor surface can increase micromotion, fibrous tissue formation, bacterial attachment, or delayed endothelial healing.
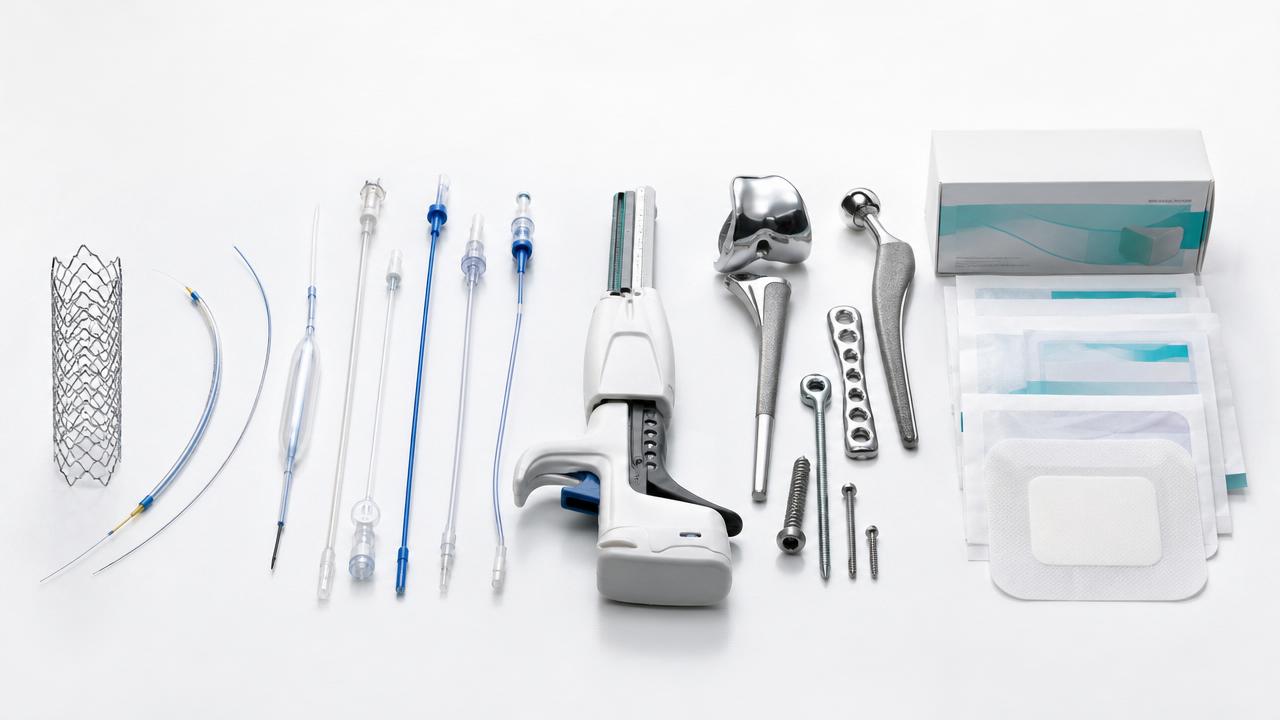
How do risks differ by application, such as joints, stents, catheters, or spinal implants?
Different implants face different biological and mechanical challenges.
That means implantable biomaterials must be judged in context, not by reputation alone.
Orthopedic and joint replacement
These devices face high repetitive loads and long service life demands.
Long-term risk often comes from wear debris, loosening, poor bone ingrowth, or mismatch between stiffness and surrounding bone.
Porous titanium and optimized bearing materials often improve fixation and reduce revision pressure.
Cardiovascular implants and stents
Blood-contact devices must resist thrombosis, corrosion, and inflammatory reactions.
In this setting, surface chemistry, coating stability, and endothelial compatibility are as important as radial strength.
Poor implantable biomaterials selection may contribute to restenosis, clot risk, or late device failure.
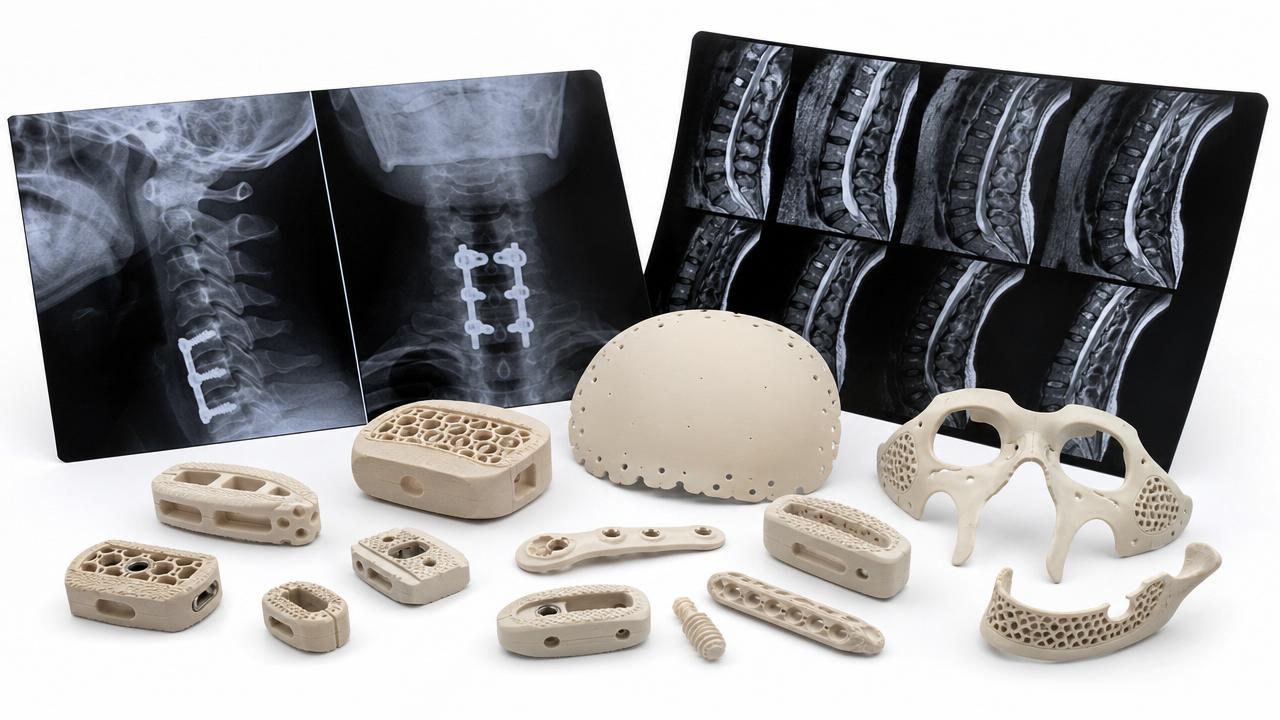
Spinal cages and fixation systems
Here, imaging visibility, stiffness balance, and fusion support are major concerns.
PEEK offers radiolucency, while titanium often supports stronger osseointegration.
The wrong tradeoff may increase subsidence, nonunion, or later instability.
Catheters and minimally invasive consumables
Polymer flexibility is useful, but long-term contact with blood or tissue raises coating durability and thrombogenicity questions.
If the material sheds particles or loses surface function, complications may follow.
How can you compare safer implantable biomaterials before a procedure?
A safer choice usually comes from structured comparison rather than brand familiarity.
Focus on evidence, not marketing language.
Useful questions include the following:
- How long has this material been studied in this exact implant type?
- What are the known failure modes after five years or more?
- Does the surface support bone, blood vessel, or soft tissue healing?
- Is there a known allergy, corrosion, or wear concern?
- What revision challenges appear if the implant fails later?
What common myths lead to poor implantable biomaterials decisions?
Several assumptions can hide real long-term risk.
Myth 1: The strongest material is always the safest
Very stiff materials may shift stress away from natural bone or tissue.
That can weaken surrounding structures over time.
Myth 2: If a material is approved, all versions perform equally
Approval does not erase differences in design, surface finishing, cleanliness, or manufacturing consistency.
Two implants using similar implantable biomaterials may still perform differently.
Myth 3: Newer automatically means better
Innovation is valuable, but long-term evidence takes time.
A promising biodegradable or coated material may still have limited real-world follow-up.
Myth 4: The implant material matters more than the whole system
Procedure technique, implant geometry, patient biology, and postoperative care all interact with implantable biomaterials.
The best material can still underperform in the wrong system.
What practical steps reduce long-term risk when evaluating implantable biomaterials?
A practical review process helps convert technical information into better decisions.
- Match the material to the body site and mechanical demand.
- Check whether long-term data exist for that exact indication.
- Look for biological safety testing and surface validation.
- Consider allergy history, bone quality, healing status, and blood-contact conditions.
- Compare revision complexity, not only first-procedure convenience.
- Review whether cost savings today could increase failure costs later.
This is especially important in high-value consumables where short-term price pressure may hide long-term clinical tradeoffs.
Across orthopedic implants, cardiovascular devices, and advanced polymer systems, durable compatibility remains the core priority.
FAQ summary: how should implantable biomaterials be judged?
Implantable biomaterials shape healing quality, device durability, and revision risk long after surgery ends.
The safest option is rarely the cheapest, newest, or strongest by description alone.
It is the one supported by biological compatibility, sound engineering, and real long-term evidence in the intended application.
Before any implant procedure, compare implantable biomaterials carefully, ask targeted questions, and review outcome data beyond the operating room.
That extra diligence can reduce avoidable complications and support safer recovery for years ahead.
Get weekly intelligence in your inbox.
No noise. No sponsored content. Pure intelligence.
News Recommendations