Global Implant & Medical Consumables Systems
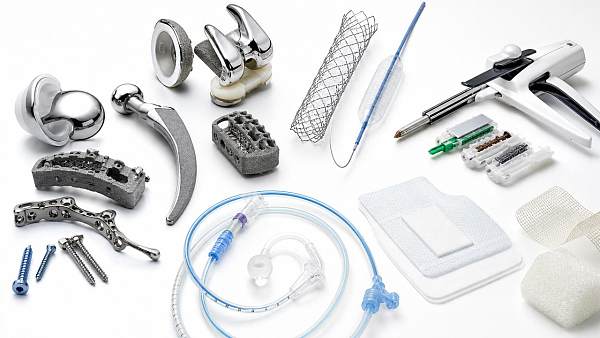
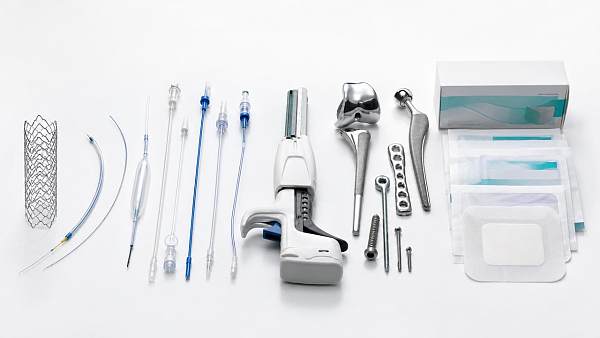
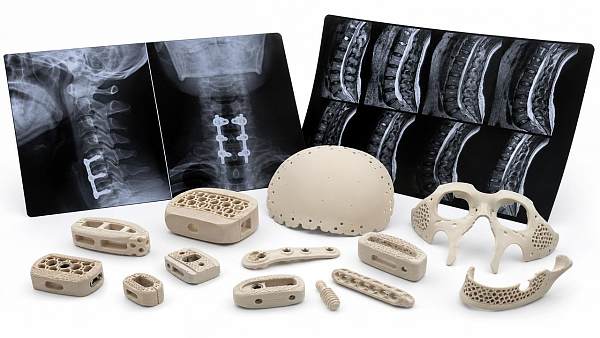
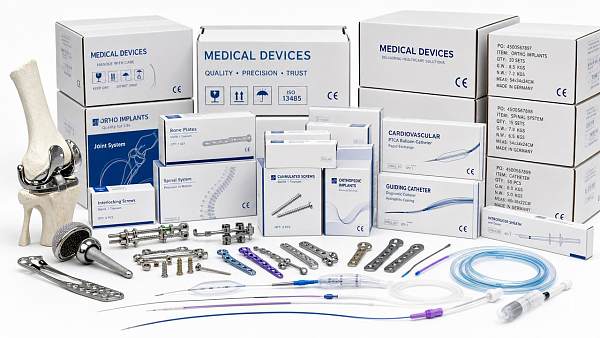
The Global Implant & Medical Consumables Systems (IMCS) is a premier intelligence portal dedicated to orthopedic replacement implants, cardiovascular interventional devices, minimally invasive surgical consumables, and high-end tissue regeneration materials.
Our Story
IMCS is a premium global intelligence platform focusing on high-end medical implants and consumables. Covering orthopedics, cardiovascular intervention, minimally invasive surgery, polymer catheters and wound care, it connects advanced material biocompatibility, precision manufacturing with Class III device regulations and VBP policies. Backed by a professional think tank, IMCS empowers medical manufacturers with clinical validation, regulatory compliance and cost-control strategies to accelerate industrial upgrading.
Core Milestones
Driven by trauma treatment demands. Basic metal materials such as stainless steel were applied in human implantation. The first hip replacement and implantable pacemaker laid the foundation for modern medical consumables.
The concept of biocompatibility was established. Titanium alloy and medical polymers were widely used. PTCA, bare metal stents and laparoscopic staplers came into clinical use. ISO 10993 and EU medical device regulations took initial shape.
DES, PEEK materials, porous trabecular structure, hydrophilic-coated catheters and silver-ion dressings were fully industrialized. The five major product sectors of IMCS were completely formed. High-precision micromachining became the core industry threshold.
Worldwide medical cost control promoted volume-based procurement. Severe price cuts occurred in stents and orthopedic implants. CE MDR enforced strict CER clinical evaluation for Class III medical devices. Manufacturers shifted from low-price competition to high-value innovation.
Focus on bionic 3D printing, biodegradable polymers and ultra-minimally invasive intervention such as TAVR. The industry pursues long-term in-vivo safety & human-body integration. IMCS serves as an elite think tank covering toxicology verification, clinical compliance and VBP capital simulation.
Latest Article